6 Label Tips for Cosmetics and Beauty Products
⚞ The Highlights:
- Cosmetics labels in the U.S. must follow FDA rules under the FD&C Act, plus the additional requirements added by MoCRA (Modernization of Cosmetics Regulation Act of 2022).
- Required elements include the product name, identity statement, net quantity, ingredient list (in descending order), name and place of business, country of origin, and any required warnings.
- The right adhesive matters as much as the design. Bathroom and shower environments call for high-solvent-resistance adhesives that can handle moisture, alcohol, and humidity.
- Premium finishes (foil, embossing, soft-touch lamination, spot UV) are how beauty brands signal quality on the shelf without saying a word.
Cosmetic and beauty product labels carry more responsibility than most product labels. They have to meet federal labeling rules, hold up to moisture and frequent handling, and earn a buyer’s eye in a category where the package often sells the product. Below are six things we tell brands to think about when designing or refreshing a cosmetic label, with current FDA framework first and design and material decisions after.
1. Follow the FDA’s cosmetic label guidelines (and the new MoCRA requirements)
A cosmetic label that doesn’t follow federal rules can be considered “misbranded” under the Federal Food, Drug, and Cosmetic Act (FD&C Act) and is subject to FDA enforcement. Two layers of regulation apply:
- Long-standing FDA cosmetic labeling rules under 21 CFR 701, 740, and 700.25 (covering principal display panel, ingredient declaration, warnings, and tamper-resistant packaging).
- The Modernization of Cosmetics Regulation Act (MoCRA), signed into law December 29, 2022. The largest update to U.S. cosmetics regulation in decades. MoCRA adds federal requirements around facility registration, product listing, adverse event reporting, safety substantiation, and additional labeling elements (including fragrance allergen disclosure on a phased timeline).
If you’re updating a label in 2026, you need to be current on both layers. The FDA’s cosmetics guidance pages are the authoritative source. Bookmark them.
What counts as a “cosmetic” product?
Under the FD&C Act, cosmetics are products applied to the body for aesthetic purposes that don’t alter the body’s structure or function (soaps that only claim to cleanse are excluded). The category includes skin creams, lotions, perfumes, lipsticks, nail polishes, eye and facial makeup, shampoos, permanent waves, hair colors, toothpastes, deodorants, and any material intended for use as a component of a cosmetic.
Some products serve both cosmetic and therapeutic purposes (fluoride toothpaste, sunscreen, anti-dandruff shampoo, antiperspirant deodorants). Those are regulated as both cosmetics and drugs, which means more stringent requirements including FDA registration and adherence to drug manufacturing practices.
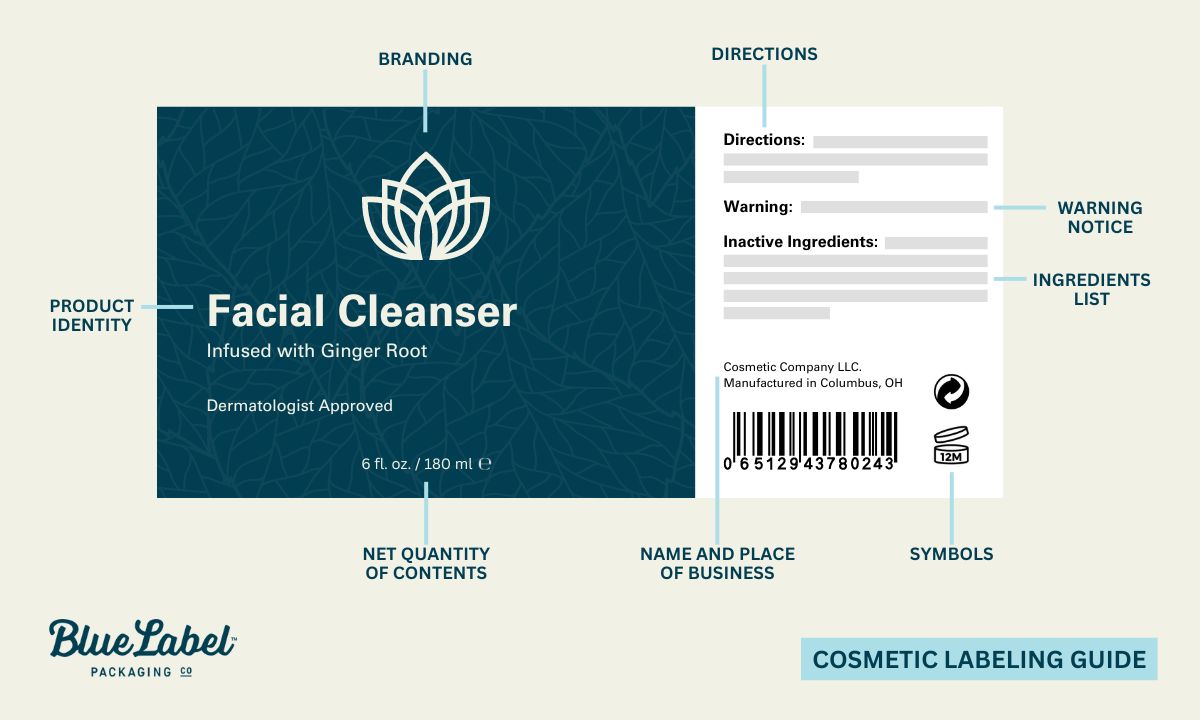
FDA cosmetic labeling guidelines
The FDA’s cosmetic labeling guide is the document that walks through the specifics. The required label elements:
- Label statements required by the FD&C Act must be visible on both inside and outside containers or wrappers.
- Ingredient listing and net quantity statement are required only on the outer container’s principal display panel (per 21 CFR 701 and 740).
- The principal display panel must show the product name, descriptive nature or use, and accurate net quantity of contents (in weight, measure, or numerical count). Net quantity declaration sits at the bottom in a type size regulated by container size.
- Solid, semi-solid, or viscous cosmetics are declared in pounds/ounces; liquids in U.S. gallons/quarts/pints/fluid ounces. Quantities greater than one pound or one pint should also state largest whole units, with metric measurements optional.
- Name and place of business of the firm marketing the product goes on an information panel: street address (unless listed in current directories) plus city, state, and zip code. If you’re not the manufacturer, indicate “Manufactured for…” or “Distributed by…” before the company name.
- Country of origin must appear on imported articles.
Declaration of ingredients
Cosmetics for retail sale must include an ingredient list. Professional-use products are exempt unless sold to consumers (and MoCRA tightened the labeling required on professional-use products specifically. Verify current rules with FDA before relying on the exemption).
- The ingredient declaration must be conspicuous and readable at purchase. It can appear on any information panel, or on an affixed tag/tape/card if the package design doesn’t allow it on the container.
- Letter height: minimum 1/16 inch on most packages; minimum 1/32 inch for packages under 12 square inches.
- Off-package labeling is allowed in specific cases (for example, cosmetics in compartmented trays without a folding carton).
- Ingredients listed in descending order of predominance, with two exceptions: color additives and ingredients at 1% or less can be listed in any order. Names must follow regulation-established terms; some can be grouped as “and other ingredients.”
- For cosmetics also classified as drugs, drug ingredients are labeled first as “active ingredient(s).”
Label warnings
Cosmetics that could be hazardous if misused must carry clear warning labels and directions for safe use under 21 CFR 740. This applies especially to aerosol cosmetics, feminine deodorant sprays, and children’s bubble baths.
Manufacturers aren’t required by the FD&C Act to test products for safety, but the FDA strongly encourages it. If safety hasn’t been substantiated, the label must include a specific warning indicating the product’s safety has not been determined.
Tamper-resistant packaging
Liquid oral hygiene and cosmetic vaginal products sold at retail must be in tamper-resistant packaging, with a distinctive indicator or barrier that signals tampering to the consumer. The package must clearly display a statement about its tamper-resistant feature, which has to remain visible even if the feature is compromised (per 21 CFR 700.25).

2. Highlight features and benefits that buyers actually look for
The required information is the floor, not the ceiling. The claims you choose to feature on the front of the label often decide whether a buyer picks up your product. The category-specific claims that carry weight in beauty:
- Organic: signals naturally sourced ingredients to consumers seeking chemical-free options.
- Vegan: communicates no animal-derived ingredients, which matters to ethical and environmentally conscious shoppers.
- Cruelty-free / not tested on animals: signals that neither the product nor its ingredients were tested on animals.
- Alcohol-free: attracts buyers looking for gentle formulations that won’t dry out or irritate sensitive skin.
- Hypoallergenic: suggests a lower risk of allergic reaction, important for sensitive-skin shoppers.
If any of these claims are truthful and not misleading, they can go on your label. The FDA’s general rule is that claims must be substantiated; some terms (like “organic” or “natural”) have specific definitions tied to USDA or other agency standards, so verify before claiming.
Expiration dates: U.S. law doesn’t currently require expiration dates on cosmetics, but each company is responsible for the safety of its products. If your products have a meaningful shelf life, including the date is good practice, both for customer trust and for liability.
3. Plan label shapes and sizes for every container in your line
Unless you sell a single product, your labels probably aren’t a one-size-fits-all design. Common cosmetic containers include:
- Airless bottles
- Pumps
- Sprayers
- Jars
- Tubes
- Droppers
- Compacts
Each one calls for a different label shape and size. You can keep your color scheme, font, and visual identity consistent across the full line, but you’ll need to adapt the layout, the required information, and any premium design elements to fit each container’s surface.
4. Choose the right printing techniques and materials
Beauty and cosmetic labels are where premium printing techniques earn their keep. The right finish or technique can make the difference between a product that looks expensive and one that looks generic.
Cosmetic finish comparison
| Finish or technique | What it does | Best for |
|---|---|---|
| Lamination (gloss, matte, soft-touch) | Adds a clear protective layer over the label. Soft-touch lamination feels velvety; gloss adds shine; matte mutes shine. | Premium serums, perfumes, anything that needs a tactile cue of quality. Soft-touch is the most-requested finish in luxury beauty. |
| UV varnish | Coats the label with a thin layer of cured liquid for protection. Available as full-coverage or “spot UV” (selectively applied). | Adding a pop of gloss to specific design elements (logo, product name) without coating the whole label. |
| Hot foil stamping | Applies metallic or specialty foil to specific areas. Gold, silver, copper, and holographic options. | Logo accents, brand names, or borders. The signature look for premium and luxury beauty. |
| Embossing / debossing | Raises (emboss) or recesses (deboss) parts of the label for a tactile effect. | Brand names, logos, monograms. Adds a hand-feel that flat printing can’t match. |
| Double-sided labels | Printed on both sides so the back of the label shows through a clear container. | Clear bottles where you want decorative imagery or ingredient information visible from inside. |
| Waterproof film labels | Vinyl or polypropylene labels that hold up to water, humidity, and frequent handling. | Anything used in the bathroom, shower, or refrigerator. Standard for most beauty SKUs. |
| Hang tags | Custom tags attached to the product or package, separate from the main label. | Extra branding real estate, special promotions, gift packaging, or storytelling that won’t fit on the primary label. |
Material recommendations by product type
The product itself often dictates which label material is the right call. A few patterns we see often:
- Skincare creams and serums (jars): BOPP or polypropylene with soft-touch or matte lamination. Holds up to humidity, fingerprints don’t show as much, and the matte finish reads premium.
- Perfumes and fragrances (glass bottles): clear polypropylene with hot foil stamping or embossing. The clear material disappears against the bottle so the design feels printed directly on the glass.
- Shampoos, conditioners, body wash (squeeze bottles): waterproof BOPP with high shear-resistance adhesive. The label sits in a wet, flexed environment and needs to survive both.
- Lipsticks and small tubes: small-format polypropylene labels, often with a thin UV varnish or lamination. The container is small, so the label has to take the wear of being held, opened, and dropped.
- Nail polishes (small glass bottles): polypropylene with chemical-resistant adhesive. The cap area is exposed to acetone-based products regularly during use.
- Bar soaps: uncoated paper for a craft, natural feel; or BOPP if the bar is wrapped in a clear sleeve and the label needs to stay readable through condensation.
- Cosmetic powders and compacts: small-format film labels with a fine-detail finish (often debossing) for the brand mark, and a separate ingredients label inside the case.
5. Pick the right adhesive for the environment
An elegant cosmetic label is only useful if it stays on the product. Beauty products often live in environments that are hard on labels: humid bathrooms, hot showers, cold refrigerators, frequent handling, and occasional contact with the product itself (oils, alcohols, acetones, fragrance solvents).
Adhesive performance
Cosmetic labels need to hold up against water, alcohol, plasticizers, and other harsh substances without losing adhesion or appearance. The right adhesive doesn’t just keep the label on the package; it keeps the label looking new throughout the product’s life.
High-quality adhesive types
For products that won’t see humidity, a standard all-temperature adhesive works fine. For products that will live in a bathroom, gym bag, or refrigerator:
- High solvent resistance. Keeps the label adhering through exposure to water, alcohol, and other solvents. Standard for most beauty SKUs that touch water during use.
- High shear resistance. Keeps the label intact through constant stress without tearing or cracking. Especially important for squeeze bottles and tubes that flex during use.
Match the adhesive to the package
The container’s material affects which adhesive will work best:
- Soft-touch coated containers need a very aggressive permanent adhesive to bond properly. The velvety surface is harder for standard adhesives to grip.
- Clear film labels on glass or plastic need ultra-clear permanent adhesives so the label edge stays invisible.
- Frequently flexed packages (squeeze bottles, tubes) need higher shear resistance to handle the constant deformation.
- Refrigerated products (some serums, eye creams) need cold-temperature adhesives that don’t get brittle below 40°F.

6. Design a label people want to pick up
Walk down any beauty aisle and you’ll see the impact of label design as much as the product itself. Beauty is a visual category. The label is doing the selling. A few design choices that consistently work:
- Use color psychology deliberately. Color carries emotional cues. Pinks and pastels read soft and feminine; blacks and metallics read luxe; greens and earth tones read natural. Match the palette to what you want the buyer to feel.
- Build clear typography and visual hierarchy. The buyer should be able to find the product name, type, and size at a glance. Hierarchy matters most when the buyer is comparing similar SKUs in your line.
- Add tactile or shape variation. Embossing, foil, and unusual label shapes give the package a hand-feel. In a category where people pick up the product before deciding to buy, that physical interaction matters.
- Tell a story. The label is one of the first places a buyer encounters your brand voice. Even one line of copy that hints at the product’s origin, philosophy, or ritual can make the package feel intentional.
- Try minimalism if the category is busy. Cleaner labels get noticed in a category where most competitors over-decorate.
- Color-match the product to the label. A cohesive color story between the package and the product creates a unified visual signature.
- Test patterns as a differentiator. Abstract patterns or unusual textures can give a product an edge on shelf without losing broad appeal.
Frequently asked questions
What does a cosmetic product label have to include in the U.S.?
Required elements include the product name, identity statement (what the product is), accurate net quantity of contents, ingredient list in descending order of predominance, name and place of business of the firm marketing the product, country of origin (for imports), and any required warnings. The principal display panel and information panel both have specific layout and type-size requirements under 21 CFR 701 and 740.
What is MoCRA and how does it affect cosmetic labels?
MoCRA (the Modernization of Cosmetics Regulation Act of 2022) is the largest update to U.S. cosmetics regulation in decades. Beyond labeling, it adds federal requirements for facility registration, product listing, adverse event recordkeeping, and safety substantiation. On the labeling side, it adds new requirements including fragrance allergen disclosure on a phased timeline. If you’re updating a cosmetic label in 2026, check FDA.gov for the current MoCRA labeling requirements that apply to your product category.
Do I have to list ingredients on my cosmetic label?
Yes, for cosmetics sold at retail. Ingredients must be listed in descending order of predominance, in conspicuous and readable type. Letter height is at least 1/16 inch on most packages, or 1/32 inch on packages under 12 square inches. Color additives and ingredients at 1% or less can be listed in any order. Professional-use products have different rules, which MoCRA tightened. Verify current requirements before relying on the exemption.
What’s the best label finish for cosmetic products?
It depends on the product and the brand positioning. Soft-touch lamination is the most-requested finish in luxury beauty for its velvety hand-feel. Hot foil stamping (gold, silver, holographic) is the standard for premium accents on logos and brand names. Spot UV adds shine to specific design elements without coating the whole label. Matte lamination reads understated and premium; gloss reads bright and approachable. Most beauty brands use a combination, depending on the SKU.
What kind of adhesive should I use for shower or bathroom products?
For products used in moist or humid environments. Shampoos, conditioners, body washes, face cleansers. Use an adhesive with high solvent resistance and high shear resistance. Solvent resistance keeps the label sticking through exposure to water, alcohol, and ingredient interactions. Shear resistance keeps the label intact when the bottle is squeezed or flexed. Standard all-temperature adhesives aren’t enough for these environments.
Do cosmetic labels need expiration dates?
U.S. federal law doesn’t currently require expiration dates on most cosmetic products, but the manufacturer is responsible for product safety. If your product has a meaningful shelf life, including an expiration or “period after opening” (PAO) symbol is good practice for both customer trust and liability. The EU and other jurisdictions have stricter rules, so if you’re selling internationally, check the requirements for each market.
What materials work best for cosmetic labels?
BOPP (biaxially oriented polypropylene) and polypropylene films are the workhorses for most cosmetic labels. They’re waterproof, durable, and accept a wide range of finishes. Clear polypropylene works well on glass perfume bottles where you want the design to look printed directly on the bottle. Uncoated paper fits craft or natural-positioned products. The right pick depends on the package material, the product’s environment, and the brand aesthetic.
Ready to put this to work?
If you’re designing or refreshing a cosmetic label, we can help you sort through the format, materials, finishes, and compliance pieces. Take a look at our cosmetic labels page for an overview of what we offer, or request a sample pack to see finishes and materials in person.
If you’re still looking for a designer, our designer directory can help you find someone who’s worked on beauty packaging before. And when you’re ready, get in touch and we’ll talk through your project.




