Which Foods Are Not Required to Have a Nutrition Label?
⚞ The Highlights:
- Most packaged foods sold at retail in the U.S. require an FDA Nutrition Facts label, but six categories qualify for exemption.
- The exemptions cover raw produce and seafood, foods with insignificant nutritional value, immediate-consumption foods, very small packages, bulk foods for further processing, and qualifying small businesses.
- Even if a product is exempt from the Nutrition Facts panel, it still needs identity statement, net quantity, manufacturer info, ingredient list, and allergen declarations.
- The moment a label or marketing material makes any nutrient or health claim (“low fat,” “good source of fiber,” “heart healthy”), the full Nutrition Facts panel becomes required. Even for otherwise-exempt products.
FDA regulations require that “almost all packaged foods intended for human consumption and sold at retail must include a Nutrition Facts label.” That panel has to show calories, fat, sodium, carbohydrates, protein, and other specific nutrients per serving. But there are six well-defined exemptions, and small food brands often qualify for at least one of them.
Below is who qualifies, what disqualifies you, and what’s still required even when you’re exempt. If you’re launching a food product or refreshing your packaging, this is the framework to work through before designing the label.
The six FDA nutrition label exemptions at a glance
| Exemption type | What qualifies | What disqualifies you |
|---|---|---|
| Raw produce, fish, and seafood | Single-ingredient raw fruits, vegetables, or fish sold without claims; participates in a voluntary FDA labeling program (21 CFR § 101.45) | Any nutrient or health claim on the package; multi-ingredient products; processed products |
| Insignificant nutritional value | Plain coffee beans, tea leaves, spices, food colors, flavor extracts. Products with no meaningful calories, fat, sugar, or measurable nutrients | Adding ingredients that contribute nutrients; making any nutrient claim |
| Immediate consumption | Restaurant meals, freshly prepared deli items, foods consumed on-site | Prepackaged retail products; products sold for later consumption |
| Very small packages | Products with less than 12 square inches of label space, with no nutrient or health claims on package or marketing | Any nutrient or health claims on packaging or marketing materials, even if the package is small |
| Bulk for further processing | Ingredients shipped to manufacturers for further processing, not intended for retail sale | Retail-ready packaging; nutrition claims on the packaging |
| Small business / low volume | Companies with fewer than 100 full-time employees selling under 100,000 units annually; annual FDA filing required | Exceeding either threshold; making nutrient or health claims; failing to file the annual exemption with FDA |
Source: FDA.gov
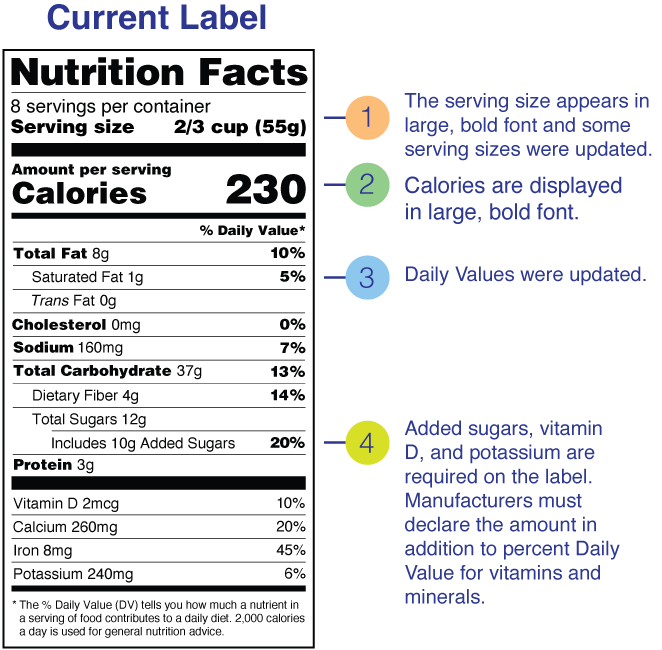
This panel must follow a standardized format, including calories, fat, sodium, carbohydrates, protein, and other required nutrients per serving.
Most foods require a nutrition label
Before walking through the exemptions, the baseline rule: most packaged foods sold at retail in the U.S. require an FDA Nutrition Facts label. The standardized panel has to display calories, fat, sodium, carbohydrates, protein, and other required nutrients per serving, in the format the FDA specifies.
The exemptions below are real but narrow. If you’re not sure whether your product qualifies, the safest assumption is that it doesn’t. And verifying with the FDA’s published guidance before you commit to packaging is worth the time.
The six exemptions in detail

1. Raw fruits, vegetables, and fish
Single-ingredient raw produce or fish sold without any nutrient or health claims fall under a voluntary FDA labeling program (21 CFR § 101.45). The program asks producers to provide nutrition information for the 20 most-consumed raw fruits, vegetables, and seafood, but compliance is voluntary, not mandatory.
The exemption applies to whole, single-ingredient items as they leave the farm or boat. Once you process, package, or combine them with other ingredients, the exemption stops applying.
2. Foods with insignificant nutritional value
Products that lack meaningful calories, fat, sugar, or measurable nutrients can be exempt. Common examples:
- Plain coffee beans (whole bean or ground, no flavorings)
- Tea leaves and tea bags (no added ingredients)
- Spices and herbs sold as single-ingredient products
- Food colors (FDA-approved color additives)
- Flavor extracts (vanilla extract, lemon extract) where nutrient content is negligible
The minute you add ingredients that contribute calories or nutrients (sugar, milk solids, oils), the exemption no longer applies.
3. Foods sold for immediate consumption
Restaurant meals and freshly prepared deli items qualify only when they’re consumed on-site or sold for immediate consumption. Examples:
- Restaurant menu items prepared and served at the restaurant
- Deli sandwiches, soups, and prepared foods served from a deli counter
- Bakery items sold on-premises
- Catering items prepared for an event
The moment you prepackage a product for retail sale (a deli sandwich in a clamshell sold at a grocery store, for example), this exemption stops applying. Some restaurant chains also have separate menu-labeling requirements under different FDA rules. That’s a different conversation.
4. Foods in very small packages
Products with less than 12 square inches of total label space can be exempt from the Nutrition Facts panel. Common examples:
- Single-serving condiment packets (ketchup, mustard, soy sauce)
- Tea bags or coffee pods sold individually
- Hard candies, gum sticks, and similar small-format products
- Spice or seasoning sample packets
The catch: the exemption only applies if the product makes no nutrient or health claims anywhere on the package or in marketing materials. The moment you say “low sodium” or “no sugar added” on the wrapper, the full Nutrition Facts panel becomes required, regardless of package size.
5. Bulk foods for further processing
Ingredients shipped to manufacturers for further processing are exempt when they’re not intended for retail sale and don’t carry nutrition claims. Examples:
- Bulk flour, sugar, or oil shipped to a bakery or food manufacturer
- Bulk spices or seasonings shipped to a co-packer
- Industrial-format ingredients in 50-pound bags or barrels
If the product is repackaged for retail sale at any point, the retailer or co-packer becomes responsible for adding the Nutrition Facts panel.
6. Small business / low volume exemption
Small food businesses can qualify for an exemption from Nutrition Facts labeling if they meet two thresholds:
- Fewer than 100 full-time employees
- Selling under 100,000 units annually of the specific product seeking exemption
The exemption isn’t automatic. Businesses must file annually through the FDA’s Small Business Nutrition Labeling Exemption system. If you make any nutrient or health claim, the exemption is void. Even if you’d otherwise qualify on the size and volume criteria.
This is the most common exemption for emerging CPG and craft food brands. If you’re under both thresholds and not making nutrient claims, file the exemption annually and you’re set.
When the exemption doesn’t apply: nutrient and health claims
The single most important catch across all six exemptions: any nutrient or health claim on the package or in marketing materials voids your exemption, regardless of which category you’d otherwise qualify under.
Examples of claims that trigger full Nutrition Facts labeling:
- Nutrient content claims: “low fat,” “high in fiber,” “no sugar added,” “good source of protein,” “fat free”
- Health claims: “supports heart health,” “may reduce risk of,” “promotes immune function”
- Structure/function claims: “supports healthy bones,” “boosts energy” (these can also trigger drug-classification concerns separately)
- Comparative claims: “less sodium than the leading brand,” “more protein than…”
If your packaging strategy depends on any of these messages, plan for the full Nutrition Facts panel from the start. Adding it later means a label redesign and a reprint.
Even if you’re exempt, you still need these label elements

Exemption from Nutrition Facts labeling doesn’t eliminate all label requirements. Every food product sold at retail in the U.S. still needs:
- Statement of identity. The common or usual name of the product
- Net quantity of contents. Weight, volume, or count, displayed on the principal display panel
- Name and address of the manufacturer, packer, or distributor
- Ingredient list if the product has more than one ingredient (in descending order by weight)
- Allergen declarations for any of the major allergens defined by FALCPA (milk, eggs, fish, crustacean shellfish, tree nuts, wheat, peanuts, soybeans) and FASTER Act (sesame, added in 2023)
Country-of-origin labeling and other product-specific rules may also apply depending on the category.
How to verify if your product needs a Nutrition Facts label
Walk through this checklist before committing to a label design:
- ☐ Is the product packaged for retail sale (not bulk, not immediate consumption)?
- ☐ Does the product contain meaningful calories, fat, sugar, or measurable nutrients?
- ☐ Does the product or its marketing make any nutrient or health claim?
- ☐ Does your business have 100 or more full-time employees?
- ☐ Do you sell more than 100,000 units of this product annually?
- ☐ Does the package have 12 square inches or more of label space?
If you answered “yes” to most of these, the Nutrition Facts panel is required. If you answered “no” to most of them, you may qualify for one of the exemptions. But verify with FDA before assuming.
Authoritative FDA resources to verify against:
- FDA Food Labeling Guide. The comprehensive FDA reference
- 21 CFR § 101.9(j). Federal exemption regulations
- FDA’s Small Business Nutrition Labeling Exemption. Annual filing for small business exemption
Frequently asked questions
What foods don’t need a Nutrition Facts label?
Six categories qualify for FDA exemption: (1) raw fruits, vegetables, and fish without claims; (2) foods with insignificant nutritional value like plain coffee, tea, and spices; (3) foods for immediate consumption like restaurant meals and deli items; (4) very small packages with under 12 square inches of label space; (5) bulk foods shipped to manufacturers for further processing; and (6) small businesses with fewer than 100 full-time employees selling under 100,000 units annually who file the FDA exemption.
Do I need a nutrition label if I’m a small business?
Possibly not, if you qualify for the FDA’s small business exemption. You need fewer than 100 full-time employees AND sell fewer than 100,000 units of the specific product per year. The exemption is not automatic. You must file annually through FDA’s Small Business Nutrition Labeling Exemption system. And if you make any nutrient or health claim on the label or in marketing, the exemption is void.
Does my product need a nutrition label if the package is very small?
Products with less than 12 square inches of total label space can be exempt from the Nutrition Facts panel. But only if no nutrient or health claims appear anywhere on the package or in marketing materials. Examples include single-serving condiment packets, individual tea bags, and small candy wrappers. The moment you add a claim like “low sodium,” the full panel is required.
Does coffee or tea need a Nutrition Facts label?
Plain coffee beans, ground coffee, tea leaves, and tea bags can qualify for the “insignificant nutritional value” exemption when sold without added ingredients. Once you add sugar, milk solids, oils, flavorings that contribute calories, or any nutrient or health claim, the exemption no longer applies and the full Nutrition Facts panel becomes required.
What’s required on a food label even if it’s exempt from Nutrition Facts?
Even with a Nutrition Facts exemption, every retail food product still needs: a statement of identity (common or usual product name), net quantity of contents on the principal display panel, name and address of the manufacturer/packer/distributor, ingredient list if multiple ingredients (in descending order by weight), and allergen declarations for the FDA’s major food allergens including sesame (added by the FASTER Act in 2023).
Do restaurant or deli foods need nutrition labels?
Foods sold for immediate consumption. Restaurant meals, freshly prepared deli items, on-site bakery products. Qualify for an exemption from Nutrition Facts labeling. The exemption applies only when the food is consumed on-site or sold for immediate consumption. Once a product is prepackaged for retail sale (a sandwich sold in a grocery store cooler, for example), it requires a Nutrition Facts label. Some larger restaurant chains also have separate menu-labeling requirements under different FDA rules.
Do nutrition label claims like “low fat” require a Nutrition Facts panel?
Yes. And this is the most common reason exemptions get voided. Any nutrient content claim (low fat, high fiber, no sugar added, fat free, etc.) or health claim (supports heart health, may reduce risk of) requires the full Nutrition Facts panel, even on otherwise-exempt products. If your packaging strategy depends on these messages, plan for the full panel from the start.
Confirm before you print
Most packaged foods at scale require Nutrition Facts labels. Confirming exemption status before you design or order a print run prevents costly reprints and launch delays. Both of which we’ve seen happen when a brand assumed it qualified for an exemption that didn’t actually apply once a “low sugar” claim went on the front of the package.
If you’ve worked through the checklist and your label is ready, take a look at our food labels for an overview of materials and finishes, or request a sample pack to see options in person. Get in touch when you’re ready for a quote. No minimums, fast turnaround, and an expert review on every project before anything goes to press.